Dr. Anand Kumar Sharma
studierte an der Universität Delhi, die er mit einem Doktortitel in Chemie verließ. Vormalig renommierter Wissenschaftler am URSC/ISRO, Bangalore, Indien. Er hält 16 Patente und war unter anderem Mitglied des External Advisory Committee of World Association of Nuclear Operators (WANO).
[email protected]
Magnesium: Resources and Production – Part 2 – Occurrence, extraction, electrolytic process and thermal Reduction
– Part 2 – Occurrence, extraction, electrolytic process and thermal Reduction
Magnesium is the ninth most abundant element in the universe by mass. It is plentifully available in the seawater (~ 0.13%) and earth’s crust (constitutes around 2.4%). Because of its strong reactivity, it does not occur in the native state, but rather it is found in a wide variety of compounds in seawater, natural brines, and rocks. By far the largest source is the oceans, though seawater consists of only about 0.13 % magnesium, it represents an almost inexhaustible source. Additionally, magnesium alloys are easily recyclable at low-energy costs without environmental pollution.
Magnesium – a versatile metal – Part 1 – The Lightest Structural Metal
Magnesium is the lightest structural metal and therefore a dream material for applications where fuel economy is a prerequisite. Applications of magnesium alloys are primarily motivated by their strength-to-weight ratio, ability to dampen shockwaves, close dimensional tolerance, and ease of fabrication. Magnesium alloys are therefore widely used in the construction of automobile, aerospace and ballistic missiles parts. Owing to their comparative strength and relatively lower cost, magnesium alloys are also seen as an alternative to conventional lightweight fibre metal laminates or carbon fibre composites. In this articles series in twelve parts, we will discuss from the extraction of magnesium to surface finishing of its products.
Chandrayaan-3 and the importance of surface technology for its success
Indias moon mission Chandrayaan-3 was launched on the 14th of July 2023 from Satish Dhawan Space Centre on the island of Sriharikota in southeast India. Chandrayaan-3 consists of a two-part configuration: Propulsion and lander modules. The lander successfully soft-landed on the 23rd of August 2023 and became the first probe to land near the lunar south pole. Ex-ISRO-scientist Dr. Anand Kumar Sharma gives insights into the Chandrayaan-3 project and includes the importance of surface finishing in the mission.
Surface Modification of Titanium– Final Part 6 – Plasma Electrolytic Oxidation (PFO): Factors Influencing & Composite Coatings
Multiple factors both intrinsic and extrinsic, influence the process and the nature and properties of the PEO coatings produced therein. The compositions of substrate materials and electrolyte are considered to be intrinsic factors which play a crucial role for the structure and composition of the coatings, while the extrinsic factors generally consist of electrical parameters, bath temperature, and processing time.
Surface Modification of Titanium – Part 5 – Plasma Electrolytic Oxidation – Mechanism & Microstructure
Plasma electrolytic oxidation (PEO) is also known as micro arc oxidation (MAO), electrolytic plasma oxidation (EPO), micro plasma oxidation (MPO), micro arc discharge oxidation (MDO), anodic spark deposition (ASD) or spark anodization (SA). This technique has emerged as one of the most effective, a rather inexpensive and environmentally benign process to fabricate well-adherent ceramic oxide layers on valve metals.
Surface Modification of Titanium - Part 4- Anodizing in Aqueous, Organic and Deep Eutectic Solvents
Anodizing is an electrochemical process that converts the surface of a metallic job into a protective metal oxide film. The process is carried out in a electrolytic cell where a job is made of an anode and an inert metal as a cathode. After applications, mechanisms and morphology of anoding in the July issue of Galvanotechnik, this part focuses on anodizing in aqueous, organic and deep eutectic solvents.
Surface Modification of Titanium – Part 3 – Anodizing: Applications, Mechanism, Morphology
Anodizing is an electrochemical process that converts the surface of a metallic job into a protective metal oxide film. The process is carried out in an electrolytic cell where a job is made of an anode and an inert metal as a cathode. When the electric current of sufficient voltage is passed through a suitable electrolyte the metal surface is converted to an adherent oxide coating which forms an integral part of the substrate [1–5].
Surface Modification of Titanium – Part 2 – Acid Etching & Electrochemical Polishing
Sandblasted large grit and acid-etched (SLA) titanium implants represent the most widely accepted, long-term clinically proven surface modification technique for biomedical implants, manufactured by either commercially pure titanium (CP-Ti) or Ti6Al4V ELI alloy. Electropolishing removes a
controlled amount of surface material from the outer layer of the object.
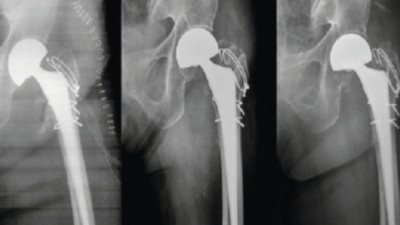
Surface Modification of Titanium – Part 1 – General Introduction & Pre-treatment
The outstanding properties of titanium and titanium alloys make them an ideal manufacturing material for the production of components for aerospace, automotive and surgical implants, among others. However, bare titanium cannot meet all functional requirements. To further improve chemical, mechanical and biological properties, the surface of titanium is modified. This article reviews recent advances in titanium alloy electrolytic surface modification methods in 6 parts.
Plating on Titanium
Plating on titanium has long been considered extremely difficult. For successful plating on titanium, the chosen process must ensure to complete removal of the native oxide before proceeding for plating. Processes of electroless nickel, gold plating, and blackening of electroless nickel on titanium alloys are discussed in the article.

![Fig. 1: Magnesium ores: magnesite, dolomite and carnallite [1]](/media/k2/items/cache/gt-2024-04-255_Generic.jpg)